IBD is a chronic, idiopathic, inflammatory disease of the digestive tract, including two phenotypes: ulcerative colitis (UC) and Crohn’s disease (CD). UC patients may experience symptoms such as bloody diarrhea, inflammation, and ulceration limited to the mucosal layer of the colon[1], while CD patients may exhibit chronic inflammatory responses throughout the digestive tract, leading to stenosis, fistulae, and intestinal obstruction.
Treatment
IBD cannot be cured, but treatments can ease symptoms and improve quality of life. Treatment options include drug therapy such as anti-inflammatory drugs, immunosuppressants, and biologics, as well as surgical resection of damaged or diseased portions of the digestive tract[2]. However, some individuals do not respond well to these treatments. Researchers have been developing new biological therapies that target key steps in known immune pathways, such as natalizumab and risankizumab. But there are still challenges, such as the risks associated with immunosuppressive therapies and individual differences in how patients respond to medication. The concept of precision medicine needs to be introduced, which further stimulates the development of more innovative drugs.
GemPharmatech‘s IBD models
Choosing the appropriate animal model for studying a new drug is a crucial first step. GemPharmatech has developed several animal models of inflammatory bowel disease (IBD), including induced models and genetically modified models of spontaneous colitis, which will aid in preclinical medication evaluation and mechanism studies.
DNBS-Induced CD models
- Aminosalicylic acid effectively alleviates DNBS-induced Crohn’s disease
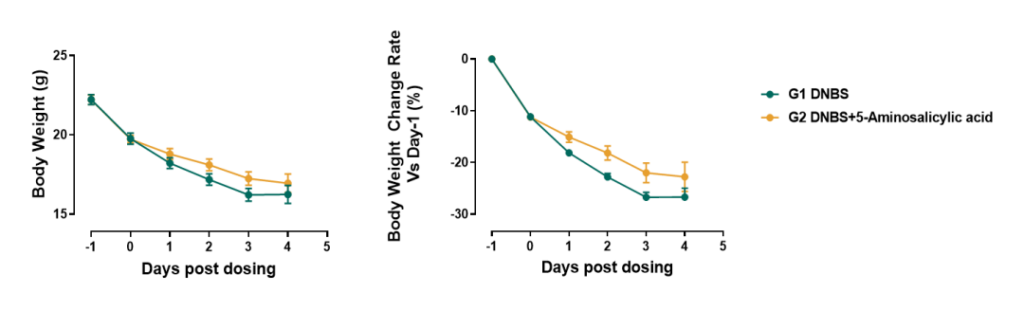
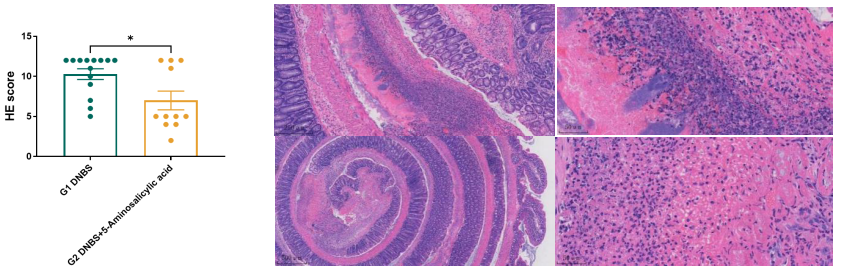
Visit our website to learn more: https://en.gempharmatech.com/content/details100032_4489252.html
Refrences:
- Ungaro, R., et al., Ulcerative colitis. Lancet, 2017. 389: p. 1756-1770.
- https://www.mayoclinic.org/diseases-conditions/inflammatory-bowel-disease/diagnosis-treatment/drc-20353320
